|
Calculate (a) the number of moles and (b) the number of grams of NH4Cl that must be added to 500mL of this solution to prepare a buffer solution with pH9.15. The treatment of other spent lithium-ion battery cathode materials, such as LiMn2O4 and LiCo1/3Mn1/3Ni1/3O2, verified the effectiveness of the method for recovering the valuable metals. The NH4Cl Chemistry A solution is 0.10M in aqueous NH3. A possible reaction mechanism between LiCoO2 and NH4Cl is proposed based on thermodynamic, X-ray diffraction, and X-ray photoelectron spectroscopy analyses. The results showed that more than 99% Co and Li were recovered under optimum conditions: temperature of 350 ☌, reaction time of 20 min, and a LiCoO2/NH4Cl mass ratio of 1:2. The reaction parameters, including roasting temperature, time, and LiCoO2/NH4Cl mass ratio, were investigated in detail. Molar Mass of NH4Cl (Ammonium chloride) Finding out the Molar mass of any molecule is relatively easy, given you know the molar mass of individual atoms. 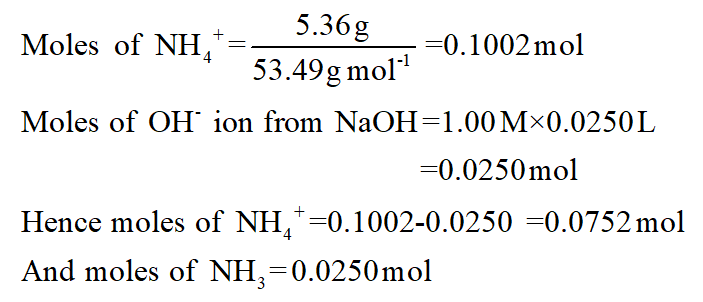
These chloride salts are readily soluble in water. L Secondary ion mass spectrum of ammonium chlo- ride cluster ions obtained by bombardment with. Convert grams NH4Cl to moles or moles NH4Cl to grams Molecular weight calculation: 14.0067 + 1.007944 + 35. Hello Guys Finding out the Molar mass of any molecule is relatively easy, given you know the molar mass of individual atoms in it. The modified Avrami equation and the MO approach were found to successfully describe the non-isothermal crystallization process. Its easy to find information of nh4cl molar mass you should know on. During this process, Li and Co are converted to chlorides due to the release of HCl and NH3 from the reaction of NH4Cl, with LiCoO2. Molecular weight of NH4Cl NH4Cl molecular weight Molar mass of NH4Cl 53.49146 g/mol This compound is also known as Ammonium Chloride. The crystallization kinetics of poly(3-hydroxybutyrate) (PHB), PHB/NH4Cl composites, PHB/ethylene-vinyl acetate (EVA 80) blends and PHB/EVA 80/1 mass NH4Cl blends were studied under non-isothermal conditions. A variety of articles about nh4cl molar mass have been classified well. The separated cathode material is roasted with NH4Cl as a fluxing agent. A novel and environmentally friendly process for utilizing spent lithium-ion batteries based on salt roasting is proposed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
