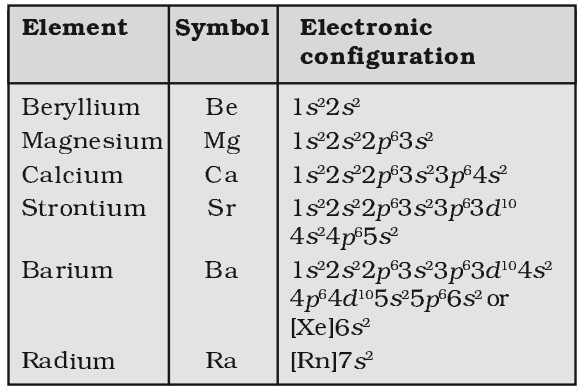
The most important commercial sources of beryllium and its compounds are beryl and bertrandite. Precious forms of beryl are aquamarine and emerald. Bussy independently isolated the metal in 1828 by reacting potassium and beryllium chloride.īeryllium is an essential constituent of about 100 out of about 4000 known minerals, the most important of which are bertrandite (Be 4Si 2O 7(OH) 2), beryl (Al 2Be 3Si 6O 18), chrysoberyl (Al 2BeO 4), and phenakite (Be 2SiO 4). This element was discovered by Louis Vauquelin in 1798 as the oxide in beryl and in emeralds. The name beryllium comes from the Greek beryllos, beryl, from Prakrit veruliya, from Pāli veuriya possibly from or simply akin to a Dravidian source represented by Tamil veiruor, viar, "to whiten, become pale." At one time beryllium was referred to as glucinium (from Greek glykys, sweet), due to the sweet taste of its salts. The speed of sound in beryllium (12,500m/s) is greater than in any other element. At standard temperature and pressures beryllium resists oxidation when exposed to air (although its ability to scratch glass is probably due to the formation of a thin layer of the oxide). It is highly permeable to X-rays, and neutrons are liberated when it is hit by alpha particles, as from radium or polonium (about 30 neutrons/million alpha particles). It has excellent thermal conductivity, is nonmagnetic and resists attack by concentrated nitric acid. The modulus of elasticity of beryllium is approximately 1/ 3 greater than that of steel. A bivalent element, beryllium is a steel grey, strong, light-weight yet brittle, alkaline earth metal, that is primarily used as a hardening agent in alloys (most notably beryllium copper).īeryllium has one of the highest melting points of the light metals. 
Beryllium ( IPA: /bəˈrɪliəm/) is the chemical element in the periodic table that has the symbol Be and atomic number 4.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
